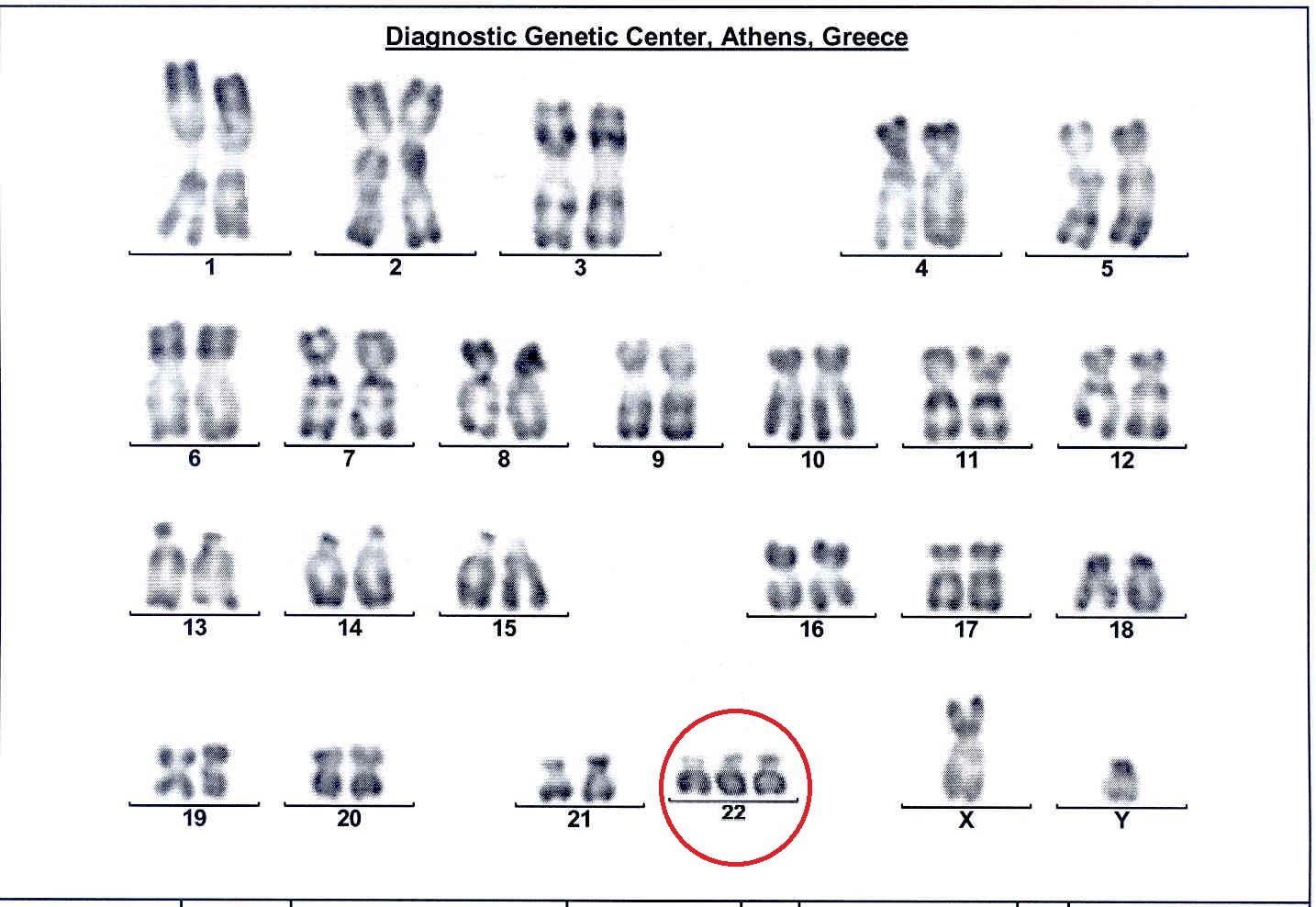
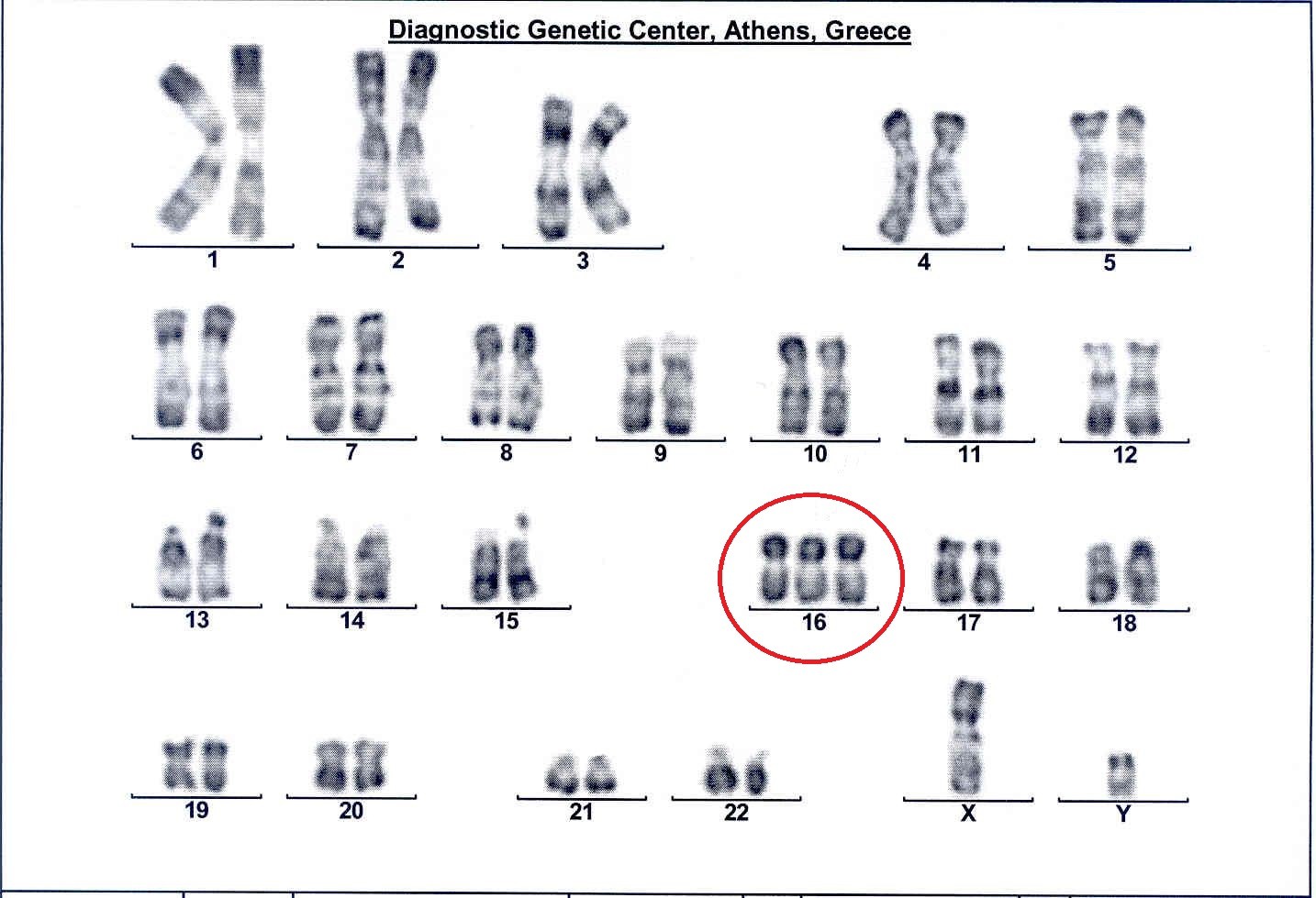
This test is particularly useful for the detection of any chromosomal aberrations that are the cause for ~60% of pregnancy loss in the 1st trimester and of 20% in the 2nd trimester. Greater than 95% of these chromosomal abnormalities in abortuses are numerical abnormalities (an extra copy of a chromosome-trisomy), which appear suddenly in the family and are therefore sporadic, and in these cases both parents have a normal karyotype. In the remaining 5% of cases, a pathological structural chromosomal abnormality is detected in the fetus, usually inherited from an unsuspecting healthy parent-carrier. If a chromosomal abnormality is detected in the abortus, then the cause of pregnancy loss has been determined, any other non-genetic investigation of the couple is of minor importance and unnecessary referrals for other time-consuming and costly tests is avoided. The value of karyotype analysis of all products of abortion is therefore obvious.
It is worth noting that the tissue obtained from the products of abortion may not be cultured, due to decomposition of the cells as a consequence of fetal death and it may therefore be impossible to perform karyotype analysis. In order to compensate for this eventuality, in such cases we apply a specially adapted QF-PCR test for chromosomes 13, 15, 16, 18, 21, 22, X and Y, which allows the detection of numerical chromosomal abnormalities (aneuploidy) of these chromosomes, which are the most common causes of miscarriages.
The difficulties and failures associated with culture may be overcome through application of molecular karyotype-aCGH of products of abortion, a test which does not require prior cell culture.
If the karyotype is found to be that of a normal female, we also perform, free of charge, further DNA analysis for the exclusion of maternal cell contamination and to ensure that the karyotype truly reflects the aborted fetus. Therefore, a small amount of peripheral blood from the mother is requested to accompany each spontaneous abortion sample.
Karyotype analysis of spontaneous abortion samples is preferably carried out from chorionic villi tissue through a sampling process which takes care to minimize maternal contamination.